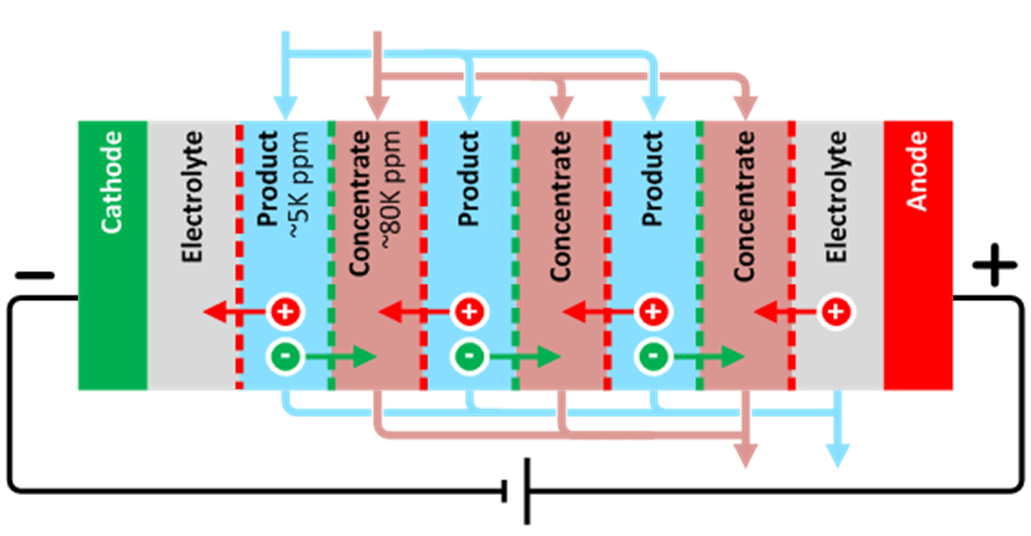
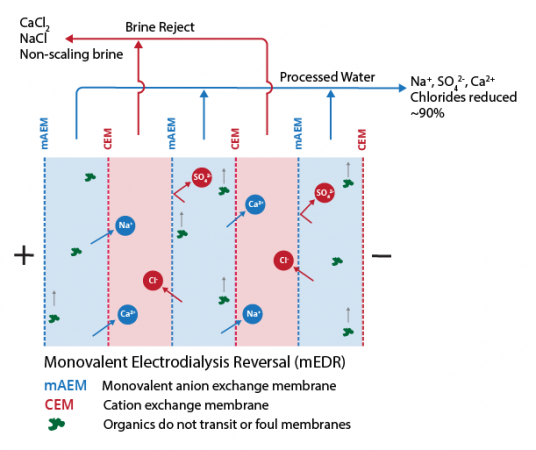
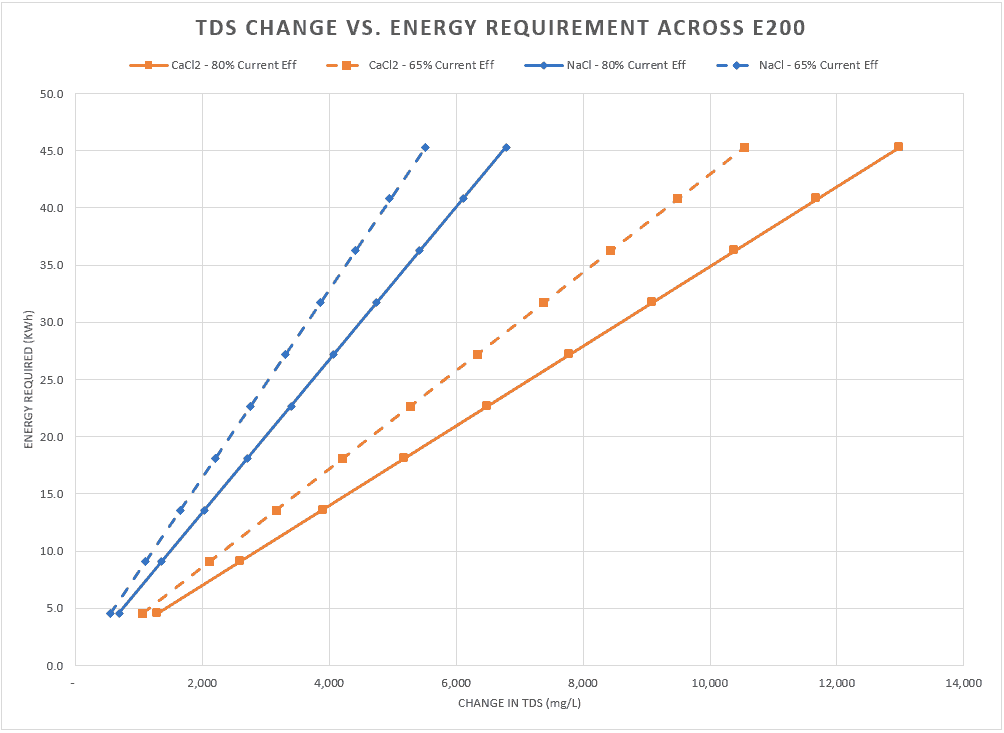
Recycling Wastewater with FlexEDR for a Chilean Heavy Industry Client
Saltworks’ FlexEDR electrodialysis reversal technology has been selected by a heavy industry client located in Chile to recycle their wastewater. The FlexEDR system, provided in partnership with Chile’s VIGAflow, will use Saltworks’ proprietary IonFlux ion exchange membranes.