First-of-Its-Kind Coal FGD Wastewater Treatment Pilot Deployed
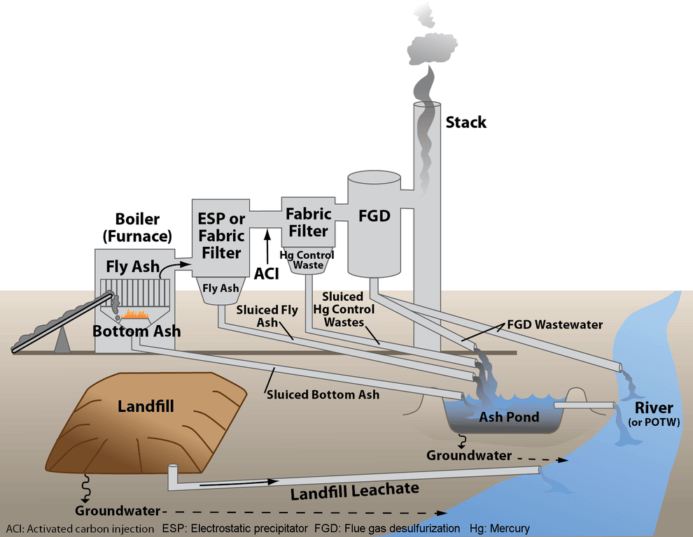
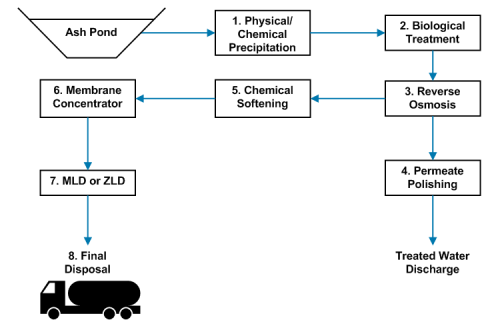
Saltworks has shipped a novel FlexEDR system to the southern United States that will clean up coal fired power wastewater. Flue gas desulfurization (FGD) wastewater is a byproduct of sulfur scrubbers, installed to prevent acid rain from coal plants.