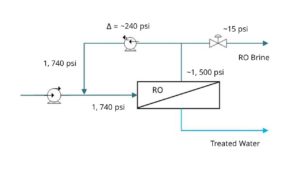
Protect Disposal Wells, Optimize Capacity, and Reduce Disposal Costs
Disposal wells are an established method for disposing of hazardous fluids by injecting them deep underground. Commingling (mixing) incompatible fluids in a disposal well may inadvertently cause solids to precipitate, which risks plugging the well. Plugging is disruptive and costly to fix, but operators can prevent it with advanced sensors, controls, and targeted wastewater treatment.